Comprehensive Study Material: Heme, Cholesterol, and Carbohydrate Metabolism
Source Information: This study material has been compiled from a lecture audio transcript and various copy-pasted text sources (personal notes, PDF/PowerPoint texts).
Introduction
This study guide explores the fundamental biochemical pathways and clinical significance of heme, cholesterol, and carbohydrates. These molecules are vital for energy production, structural integrity, signaling, and overall cellular function. Understanding their synthesis, degradation, and regulation is crucial for comprehending human physiology and disease.
I. Heme Metabolism
Heme is a critical prosthetic group found in various proteins, essential for oxygen transport, electron transfer, and drug metabolism.
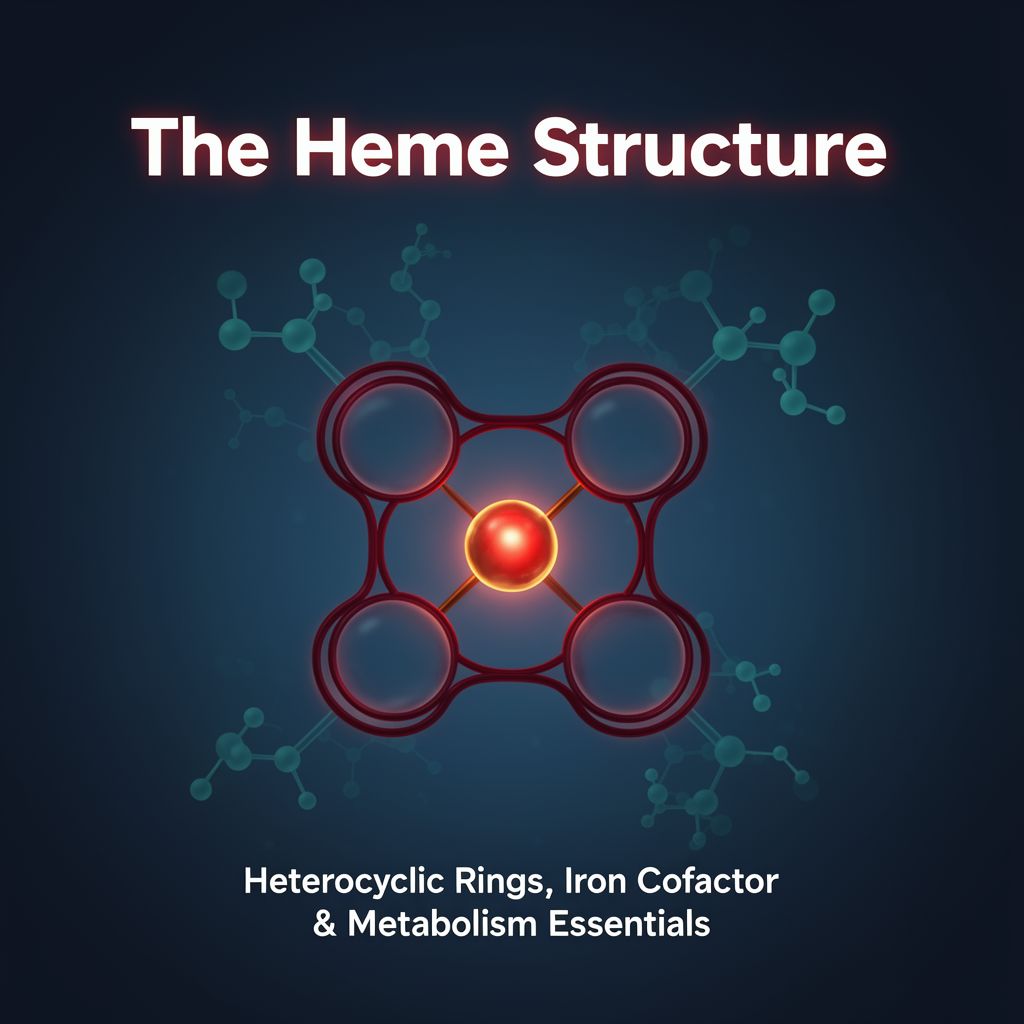
A. Heme Structure & Functions 📚
Heme features a heterocyclic ring structure composed of four pyrrole rings joined by methenyl bridges. A metal atom is chelated at its center to the nitrogen atoms of the pyrrole units.
- In Heme: The central metal atom is iron (Fe).
- In Chlorophyll: The central metal atom is magnesium (Mg).
Key Functions of Heme: ✅ Oxygen transport (hemoglobin) and storage (myoglobin) ✅ Prosthetic group for cytochrome P450 enzymes ✅ Iron reservoir ✅ Electron shuttle in the electron transport chain (cellular respiration) ✅ Signal transduction (e.g., circadian rhythms, antioxidant response) ✅ Cellular differentiation and proliferation ✅ Nitric oxide synthase activity
B. Heme Synthesis Pathway 🧬
Heme synthesis primarily occurs in the liver and bone marrow, specifically in erythroid cells (not mature red blood cells).
- Initial Step (Mitochondria): 1️⃣ Succinyl-CoA + Glycine → S-Aminolevulinate (ALA)
- Catalyzed by d-aminolevulinic acid synthase (ALA synthase).
- Involves condensation and decarboxylation.
- ALA is then exported to the cytoplasm.
- Cytoplasmic Steps:
- 2️⃣ 2 ALA → Porphobilinogen (PBG) + 2 H₂O
- Catalyzed by ALA dehydratase (also Porphobilinogen Synthase).
- Highly sensitive to heavy metal inhibition (e.g., lead poisoning).
- 3️⃣ 4 PBG → Linear Hydroxymethylbilane (HMB)
- Catalyzed by Porphobilinogen Deaminase (Hydroxymethylbilane Synthase), releasing 4 ammonia molecules.
- 4️⃣ HMB → Uroporphyrinogen III → Coproporphyrinogen III
- Uroporphyrinogen Decarboxylase converts Uroporphyrinogen III to Coproporphyrinogen III, which enters the mitochondria.
- 2️⃣ 2 ALA → Porphobilinogen (PBG) + 2 H₂O
- Mitochondrial Steps (Final):
- 5️⃣ Coproporphyrinogen III → Protoporphyrinogen IX → Protoporphyrin IX
- Involves oxidation steps catalyzed by enzymes like Protoporphyrinogen IX Oxidase.
- 6️⃣ Protoporphyrin IX + Fe²⁺ → Heme
- Catalyzed by Ferrochelatase.
- 5️⃣ Coproporphyrinogen III → Protoporphyrinogen IX → Protoporphyrin IX
C. Regulation of Heme Synthesis ⚖️
The rate-limiting step is controlled by ALA synthase.
- Feedback Inhibition: Heme directly inhibits ALA synthase activity.
- Transport Inhibition: Heme blocks ALA synthase transport from cytoplasm to mitochondria.
- Gene Expression: Heme suppresses the synthesis of ALA synthase.
- Erythroid Cells: Heme stimulates globin chain synthesis, and erythropoietin stimulates heme production, crucial for erythroblast maturation.
D. Heme Catabolism & Bilirubin ♻️
Heme cannot be reused; it is degraded, but its iron is preserved.
- Process: Aged red blood cells (~120 days) are phagocytized by macrophages in the spleen, liver, and bone marrow. Hemoglobin is split into globin and heme.
- Heme Degradation:
- 1️⃣ Heme → Biliverdin + CO + Fe²⁺
- Catalyzed by Heme Oxygenase (microsomal enzyme), requiring O₂ and NADPH.
- 2️⃣ Biliverdin → Bilirubin
- Catalyzed by Biliverdin Reductase.
- 1️⃣ Heme → Biliverdin + CO + Fe²⁺
- Bilirubin Transport & Excretion:
- Bilirubin (unconjugated/indirect) binds to serum albumin and travels to the liver.
- In hepatocytes, it's conjugated with UDP-glucuronic acid by UDP-glucuronyltransferase to form water-soluble bilirubindiglucuronide (conjugated/direct).
- Conjugated bilirubin is secreted into bile.
- In the intestine, bacterial flora convert it to urobilinogen.
- Urobilinogen is mostly converted to stercobilin (colors feces brown) or reabsorbed and excreted as urobilin in urine (yellow pigment).
E. Clinical Aspects of Heme Metabolism 🏥
- Porphyrias: Genetic disorders due to enzyme deficiencies in heme synthesis, leading to accumulation of heme precursors.
- Acute Intermittent Porphyria: Mutation in Hydroxymethylbilane Synthase, accumulating ALA and PBG.
- Porphyria Cutanea Tarda: Deficiency in Uroporphyrinogen Decarboxylase.
- Lead Poisoning: Lead inhibits several heme synthesis enzymes, notably ALA dehydratase and Ferrochelatase.
- Jaundice (Hyperbilirubinemia): Yellow discoloration of skin/sclerae (bilirubin > 2 mg/dL).
- Pre-hepatic (Hemolytic): Excess indirect bilirubin (e.g., acute hemolytic anemia).
- Hepatic (Hepatocellular): Both direct and indirect bilirubin (e.g., liver damage).
- Post-hepatic (Cholestatic): Excess direct bilirubin (e.g., bile duct obstruction).
II. Cholesterol Metabolism
Cholesterol is a vital lipid, serving as a structural component of cell membranes and a precursor for essential biomolecules.
A. Cholesterol Structure & Functions 🔬
- Structure: Perhydro-cyclo-pentano-phenanthrene (four fused rings) with a hydroxyl group at C3, a double bond at C5-C6, an 8-carbon side chain at C17, and methyl groups at C10 and C13. It has a polar head, rigid ring system, and non-polar tail.
- Functions: ✅ Cell membrane fluidity and integrity (especially nervous cells). ✅ Precursor for bile salts (fat digestion). ✅ Precursor for steroid hormones (sex hormones, stress hormones). ✅ Precursor for fat-soluble vitamins (D, K, A, E).
B. Cholesterol vs. Triglycerides & Lipoproteins 📊
- Triglycerides: Store unused calories for energy. High levels linked to obesity, metabolic syndrome.
- Cholesterol: Builds cell membranes, produces hormones.
- Lipoproteins: Transport cholesterol in blood.
- HDL ("Good" Cholesterol): High-Density Lipoprotein. More protein than cholesterol. Transports cholesterol from tissues to liver for excretion (reverse cholesterol transport). Higher HDL = lower coronary artery disease (CAD) risk.
- LDL ("Bad" Cholesterol): Low-Density Lipoprotein. More cholesterol than protein. Deposits cholesterol in blood vessels, leading to plaque buildup. Higher LDL = increased CAD risk.
C. Cholesterol Biosynthesis Pathway 🧪
- Location: Primarily liver and intestine; occurs in cytosol and mitochondria.
- Precursor: Acetyl-CoA. Requires ATP and NADPH.
- Key Steps:
- Acetyl-CoA → HMG-CoA: Two Acetyl-CoA condense to Acetoacetyl-CoA, then react with a third Acetyl-CoA to form HMG-CoA.
- HMG-CoA → Mevalonate: Rate-limiting step, catalyzed by HMG-CoA Reductase (HMGR), requiring 2 NADPH.
- Mevalonate → Isopentenyl Pyrophosphate (IPP) & Dimethylallyl Pyrophosphate (DMAPP): Involves phosphorylation and decarboxylation. These are 5-carbon isoprene units.
- IPP + DMAPP → Squalene: IPP and DMAPP condense to Geranyl Pyrophosphate (GPP, 10C), then to Farnesyl Pyrophosphate (FPP, 15C). Two FPP molecules join to form Squalene (30C).
- Squalene → Lanosterol → Cholesterol: Squalene undergoes cyclization (via Squalene Epoxidase and Lanosterol Synthase) to Lanosterol, which is then converted to cholesterol through 19 additional reactions.
D. Regulation of Cholesterol Synthesis ⚙️
HMGR is the primary regulatory enzyme.
- Feedback Inhibition: High cholesterol inhibits HMGR activity and promotes its degradation.
- Transcriptional Regulation:
- SREBP (Sterol Regulatory Element-Binding Protein): A transcription factor that activates HMGR gene expression.
- SCAP (SREBP Cleavage-Activating Protein) & INSIG proteins: When sterol levels are high, SCAP-SREBP complex remains in the ER, bound to INSIG, preventing SREBP activation.
- Low Sterols: SCAP-SREBP complex moves to Golgi, where SREBP is proteolytically cleaved by Site-1 and Site-2 Proteases (S1P, S2P). The active SREBP fragment enters the nucleus, activating HMGR gene transcription.
- Short-Term Regulation:
- AMPK (AMP-activated protein kinase): Phosphorylates and inhibits HMGR when cellular energy (ATP) is low, reducing energy-demanding cholesterol synthesis.
- Pharmaceutical Regulation:
- Statins: Competitively inhibit HMGR, mimicking mevalonate, thereby reducing cholesterol synthesis and lowering blood cholesterol.
E. Cholesterol Transport & Esterification 🚚
- Esterification: Cholesterol is converted to cholesterol esters (more hydrophobic) for storage and transport.
- ACAT (Acyl-CoA-Cholesterol Acyltransferase): Catalyzes esterification in cells, using Acyl-CoA. ACAT1 (all tissues), ACAT2 (intestine).
- LCAT (Lecithin-Cholesterol Acyltransferase): Acts on HDL, using lecithin to esterify cholesterol, trapping it within HDL.
- Reverse Cholesterol Transport: HDL picks up cholesterol from peripheral tissues, LCAT esterifies it, and HDL delivers cholesterol esters to the liver via SR-B1 receptor and ABC transporters.
III. Carbohydrate Biochemistry
Carbohydrates are polyfunctional compounds crucial for energy, structure, and cellular communication.
A. Carbohydrate Basics & Functions 💡
- Definition: Polyhydroxyaldehydes or polyhydroxyketones. "Sugars" are water-soluble and sweet carbohydrates.
- Functions: ✅ Primary energy source (glucose). ✅ Energy storage (starch in plants, glycogen in animals). ✅ Structural components (cellulose in plants, chitin in insects, bacterial cell walls). ✅ Components of DNA and RNA. ✅ Cell communication and recognition (glycobiology, glycomics).
B. Monosaccharide Classification & Stereochemistry 🔄
- Monosaccharides: Simplest carbohydrates, primary energy source, building blocks.
- Classification by Carbon Atoms: Triose (3C), Tetrose (4C), Pentose (5C), Hexose (6C, e.g., glucose).
- Classification by Carbonyl Group: Aldoses (aldehyde at C1), Ketoses (ketone at C2).
- Stereochemistry:
- Chiral Carbon: Carbon bonded to four different groups, creating enantiomers (non-superimposable mirror images).
- D/L Configuration: Determined by the -OH group on the chiral carbon farthest from the functional group. D-form (OH on right) is most common in nature.
- Epimers: Sugars differing in configuration at only one chiral carbon (e.g., glucose and mannose are C2 epimers).
C. Ring Formation & Anomers 💍
- Cyclization: 5- and 6-carbon monosaccharides form rings in solution via intramolecular hemiacetal (aldoses) or hemiketal (ketoses) formation.
- Pyranose: Six-membered ring (e.g., glucose forms glucopyranose).
- Furanose: Five-membered ring (e.g., fructose forms fructofuranose).
- Anomers: New chiral center formed at the anomeric carbon (C1 for aldoses, C2 for ketoses) upon cyclization.
- Alpha (α) Anomer: -OH on anomeric carbon is opposite to the -CH₂OH group (below the plane in Haworth projection).
- Beta (β) Anomer: -OH on anomeric carbon is on the same side as the -CH₂OH group (above the plane).
- Mutarotation: Equilibrium between α and β anomers in solution, interconverting via the open-chain form, leading to a stable optical rotation.
D. Monosaccharide Derivatives ➕
Simple sugars can be modified by adding or changing functional groups.
- Phosphorylation: Addition of phosphate groups (e.g., glucose 6-phosphate). Traps sugars inside cells.
- Oxidation: Conversion of -OH or aldehyde groups to acids.
- Amination: Replacement of -OH with amino groups (e.g., glucosamine).
- Dehydrogenation: Removal of hydrogen atoms. These derivatives are crucial for cell recognition, signaling, and metabolism.
IV. Related Metabolic & Clinical Concepts
A. Fatty Acid Metabolism Regulation ⚖️
- Acetyl-CoA Carboxylase (ACC): Key enzyme controlling fatty acid synthesis.
- Hormonal Regulation: Insulin activates ACC (synthesis), Glucagon/Epinephrine inhibit ACC (breakdown).
- Allosteric Regulation: Citrate activates ACC (abundant energy), Palmitoyl-CoA inhibits ACC (sufficient fatty acids).
- Energy Status: AMPK phosphorylates and inactivates ACC when energy is low (high AMP).
- Fed State: Insulin promotes storage, inhibits fat breakdown, stimulates malonyl-CoA (inhibits fatty acid entry into mitochondria for oxidation).
- Starvation: Glucagon/Epinephrine activate lipase, inactivate ACC, reduce malonyl-CoA, increasing fatty acid transport into mitochondria for beta-oxidation.
B. Neurodegenerative Diseases & Metabolism (MS, ALS) 🧠
Mitochondrial dysfunction and oxidative stress are common features in neurodegenerative diseases.
- Mitochondrial Dysfunction: Problems in Krebs cycle, electron transport chain, oxidative phosphorylation lead to ATP imbalance.
- Reactive Oxygen Species (ROS) & Reactive Nitrogen Species (RNS): Highly reactive molecules causing oxidative/nitrosative stress, damaging lipids, proteins, DNA. Imbalance contributes to disease progression.
- Multiple Sclerosis (MS): Demyelinating disease. ROS/RNS contribute to myelin and oligodendrocyte damage. Mitochondrial dysfunction plays a role in progression.
- Amyotrophic Lateral Sclerosis (ALS): Motor neuron disease. Characterized by oxidative/nitrosative stress and mitochondrial dysfunction. Metabolic alterations include altered NAA levels, amino acid changes, reduced antioxidants (alpha/gamma-tocopherol, GSH), and elevated oxidative damage markers (MDA, nitrite/nitrate).