This study material has been compiled and organized from various sources, including copy-pasted text and an audio lecture transcript, to provide a comprehensive overview of dissolution types and solution concentration.
🧪 Understanding Dissolution and Solution Concentration
🎯 Introduction
This study guide explores the fundamental ways substances dissolve and how we quantify the amount of dissolved material in a solution. Understanding these concepts is crucial for various scientific and practical applications, from everyday chemistry to industrial processes.
1️⃣ Types of Dissolution
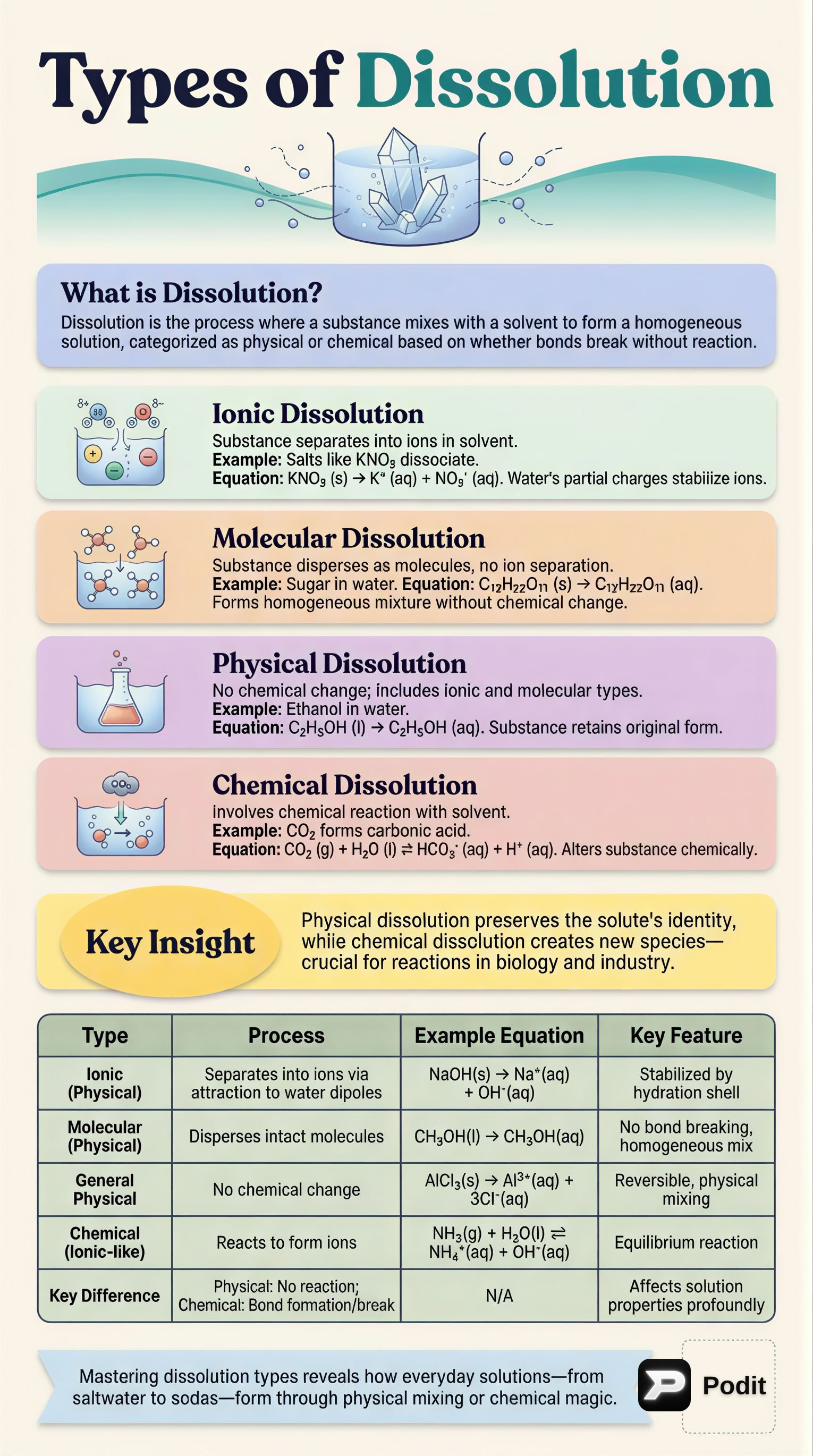
Dissolution is the process by which a solute forms a solution in a solvent. It can be broadly categorized into physical and chemical processes.
1.1 Physical Dissolution
📚 Definition: Physical dissolution is a process where a substance dissolves in a solvent without undergoing any chemical change. The chemical identity of the substance remains the same; it merely disperses within the solvent. Many ionic and molecular dissolutions are examples of physical dissolution.
1.1.1 Ionic Dissolution
📚 Definition: Ionic dissolution is the process in which an ionic compound separates into its constituent ions when dissolved in a solvent. ✅ Characteristics:
- Common for acids, bases, and salts in water.
- Water molecules, being polar, attract the cations and anions. The partially negative oxygen end (O δ–) attracts cations, while the partially positive hydrogen end (H δ+) attracts anions.
- These attractions stabilize the ions, allowing them to move away from each other and become surrounded by water molecules (hydration).
- No new chemical bonds are formed between the solute and solvent that change the solute's chemical identity.
📝 Examples:
- Potassium nitrate (KNO₃) dissolving in water:
KNO₃ (s) → K⁺ (aq) + NO₃⁻ (aq) - Sodium hydroxide (NaOH) dissolving in water:
NaOH (s) → Na⁺ (aq) + OH⁻ (aq) - Aluminum chloride (AlCl₃) dissolving in water:
AlCl₃ (s) → Al³⁺ (aq) + 3Cl⁻ (aq)
1.1.2 Molecular Dissolution
📚 Definition: Molecular dissolution is the process in which a substance disperses in a solvent in its molecular form, forming a homogeneous mixture without dissociating into ions or undergoing chemical change. ✅ Characteristics:
- Typically observed with many covalently bonded compounds.
- The molecules remain intact but are uniformly distributed throughout the solvent.
📝 Examples:
- Sugar (sucrose, C₁₂H₂₂O₁₁) dissolving in water:
C₁₂H₂₂O₁₁ (s) → C₁₂H₂₂O₁₁ (aq) - Methanol (CH₃OH) dissolving in water:
CH₃OH (l) → CH₃OH (aq) - Ethanol (C₂H₅OH) dissolving in water:
C₂H₅OH (l) → C₂H₅OH (aq)
1.2 Chemical Dissolution
📚 Definition: Chemical dissolution is the process where a substance dissolves in a solvent by undergoing a chemical interaction, leading to the formation of new chemical species. ✅ Characteristics:
- The solute reacts with the solvent, changing its chemical identity.
- Often involves the formation of new compounds or ions through a chemical reaction.
📝 Examples:
- Carbon dioxide (CO₂) dissolving in water to form carbonic acid:
CO₂ (g) + H₂O (l) ⇌ HCO₃⁻ (aq) + H⁺ (aq) - Ammonia (NH₃) dissolving in water to form ammonium and hydroxide ions:
NH₃ (g) + H₂O (l) ⇌ NH₄⁺ (aq) + OH⁻ (aq) - Sulfur dioxide (SO₂) dissolving in water:
SO₂ (g) + H₂O (l) ⇌ HSO₃⁻ (aq) + H⁺ (aq)
2️⃣ Solution Concentration
Concentration quantifies the amount of dissolved substance (solute) in a given amount of solution or solvent. Different units are used depending on the application and the amount of solute present.
2.1 Molar Concentration (Molarity)
📚 Definition: Molar concentration, or molarity (M), expresses the number of moles of solute dissolved in one liter of solution. It is a widely used unit in chemistry.
📈 Formula:
M = n / V
Where:
M= Molarity (mol/L or M)n= Number of moles of solute (mol)V= Volume of the solution (L)
💡 Insight: The symbol 'M' is read as "molar."
2.1.1 Calculating Molarity
Example 1: Calculate the molar concentration of a solution prepared with 1.25 mol of HBr acid in 0.5 L of solution.
n = 1.25 molV = 0.5 LM = 1.25 mol / 0.5 L = 2.5 M
Example 2: 3 mol of Pb(NO₃)₂ solid is dissolved in pure water to obtain a 2 L solution. What is its molar concentration?
n = 3 molV = 2 LM = 3 mol / 2 L = 1.5 M
2.1.2 Concentrated vs. Dilute Solutions
📚 Definition: These are relative terms used to compare solutions of the same substance.
- Concentrated solution: Contains a greater amount of solute per unit volume.
- Dilute solution: Contains a smaller amount of solute per unit volume. 💡 Tip: The concepts of concentrated and dilute are relative. A solution considered "concentrated" in one context might be "dilute" in another.
2.1.3 Dilution of Solutions
When solvent is added to a solution, the amount of solute (moles) remains constant, but the volume of the solution increases, thus decreasing the concentration.
📈 Dilution Formula:
M₁V₁ = M₂V₂
Where:
M₁= Initial molar concentrationV₁= Initial volume of the solutionM₂= Final molar concentrationV₂= Final volume of the solution
Example: 250 mL of 4 M NaOH solution is diluted by adding 150 mL of pure water. What is the molar concentration of the resulting solution?
M₁ = 4 MV₁ = 250 mL = 0.250 LV₂ = V₁ + added water = 250 mL + 150 mL = 400 mL = 0.400 L- Using
M₁V₁ = M₂V₂:4 M * 0.250 L = M₂ * 0.400 L1 = M₂ * 0.400M₂ = 1 / 0.400 = 2.5 M
2.2 Parts Per Million (ppm)
📚 Definition: Parts per million (ppm) is a unit of concentration used for very small amounts of dissolved substances. It refers to the mass of the substance dissolved in one liter (or one kilogram) of solution, expressed in milligrams.
1 ppm = 1 mg of solute / 1 L of solution (for aqueous solutions, assuming density ≈ 1 g/mL)
1 ppm = 1 mg of solute / 1 kg of solution
✅ Use Cases:
- Measuring trace amounts of minerals in soil or water.
- Indicating the concentration of pollutants in air or water (e.g., CO₂ in air).
- Expressing concentrations in biological samples.
📊 Examples of Concentration Units in Daily Life: | Substance | Solvent | Solute | Concentration Unit | | :-------- | :------ | :----- | :----------------- | | Lemonade | 1 L water | 100 g sugar | molarity | | Air | 1000 cm³ | 380 mg CO₂ | ppm | | Coffee | 200 mL | 40 mg caffeine | ppm | | Milk | 1 L milk | 35 g protein | molarity |
3️⃣ Check Your Understanding & Practice Problems
3.1 Types of Dissolution
-
Examine the following dissolution equations in water. Identify whether each represents ionic dissolution or molecular dissolution. a)
MgCl₂ (s) + H₂O (l) → Mg²⁺ (aq) + 2Cl⁻ (aq)b)CH₃OCH₃ (l) + H₂O (l) → CH₃OCH₃ (aq)c)HCl (g) + H₂O (l) → H₃O⁺ (aq) + Cl⁻ (aq)d)C₆H₁₂O₆ (s) + H₂O (l) → C₆H₁₂O₆ (aq) -
Examine the following dissolution equations in water. Identify whether each represents chemical dissolution or physical dissolution. a)
SO₂ (g) + H₂O (l) ⇌ HSO₃⁻ (aq) + H⁺ (aq)b)N₂O₅ (g) + H₂O (l) → 2NO₃⁻ (aq) + 2H⁺ (aq)c)Al(OH)₃ (s) + H₂O (l) → Al³⁺ (aq) + 3OH⁻ (aq)d)I₂ (s) + C₆H₆ (l) → I₂ (in benzene)
3.2 Molar Concentration Calculations
-
Calculating Molarity: a) 5.6 g of solid KOH is dissolved in pure water, and the solution volume is completed to 2 L. What is the molar concentration of this solution? (Atomic masses: H: 1 g/mol, O: 16 g/mol, K: 39 g/mol) b) How many grams of CaCl₂ are dissolved in 0.5 L of a 3 M CaCl₂ solution? (Atomic masses: Cl: 35.5 g/mol, Ca: 40 g/mol)
-
Dilution Problem: A cleaning company sells a 100 mL concentrated product containing 10 M active ingredient. How many liters of surface cleaner containing 0.1 M active ingredient can be obtained from this concentrated product?
-
Comparing Concentrations: Three NaCl solutions are prepared:
- Container 1: 0.1 mol NaCl in 1000 mL solution
- Container 2: 0.5 mol NaCl in 500 mL solution
- Container 3: 0.1 mol NaCl in 500 mL solution a) Calculate the molar concentration of the solution in each container. b) Arrange the solutions from the most dilute to the most concentrated. c) What can be done to make the concentrations of the solutions in Container 1 and Container 3 equal? Explain with calculations.
-
Real-world Application - Lemonade: A company uses 10.26 g of sucrose sugar (C₁₂H₂₂O₁₁) for 1 L of lemonade. (Molar mass of C₁₂H₂₂O₁₁: 342 g/mol) a) Calculate the molar concentration of sugar in 1 L of lemonade. b) How many moles of sugar are present in one sip (approximately 10 mL) of lemonade? c) A student tried to prepare a 1 M sucrose solution using 17.1 g of sucrose and 50 mL of water, aiming for a total volume of 50 mL. However, after dissolving, the volume was 56 mL. Is the molar concentration of the prepared solution less than or greater than 1 M? Explain the student's oversight.
-
Serial Dilution: From a 10 M sodium chloride solution, 1 mL is taken and 9 mL of water is added. Then, 1 mL is taken from this new solution and 9 mL of water is added again. This process is repeated a total of four times. Calculate the molar concentrations of the solutions after each dilution (x, y, z, and t). (Assume
V_total = V_initial + V_added_water)
4️⃣ Reflection Questions
- Can the mass of a substance with an unknown molar mass be used to calculate its molar concentration? Explain your reasoning.
- In which areas of daily life can molar concentration be used? Provide examples.
- Explain which variables should be considered to describe a solution as concentrated or dilute.
- Is there a similarity between the concepts of isotonic and hypertonic solutions (from biology) and the concepts of dilute and concentrated solutions?
💡 Conclusion
This study material has covered the essential distinctions between physical (ionic and molecular) and chemical dissolution, along with the critical concentration units of molarity and parts per million (ppm). Mastering these concepts provides a strong foundation for understanding chemical reactions and solution properties in various scientific disciplines.